EPJ E Highlight - Acanthamoeba castellanii offers a simplified model for density-driven cell migration
- Details
- Published on 24 July 2025
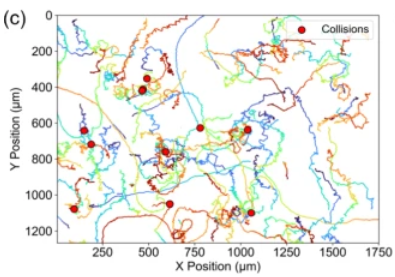
New analysis reveals that the motion of this unicellular amoeba is governed solely by cell–cell collisions, providing a useful model for isolating the effects of density on migration
Cell migration is vital to numerous biological processes. While often guided by long-range biochemical cues, it can also be influenced by direct physical collisions, which trigger biochemical signals within the affected cells. When studying this behaviour, it is important for researchers to consider how individual cell motions are affected by overall cell density. However, experiments have been complicated by the many interacting factors involved, making it difficult to isolate the specific impact of direct cell–cell collisions.
In new research published in EPJ E, a team led by Jean-Paul Rieu at Claude Bernard University Lyon 1 demonstrates how one unicellular amoeba species, Acanthamoeba castellanii (Ac), migrates in ways that are unaffected by long-range biochemical signalling – making it a promising model for studying how density influences cell migration. By using Ac, the researchers aim to gain deeper insights into the mechanisms of cell motion relevant to diverse biological processes, including immune responses, cancer cell invasion, and tissue development.
EPJ E Topical Issue: Charged Species in Bulk and at Interfaces: Interaction, Mobility, Transport, and Regulation
- Details
- Published on 06 May 2025

Guest Editors: Emanuela Bianchi, Jan K.G. Dhont, Gerhard Kahl, Kyongok Kang and Holger Stark.
The topical issue titled “Charged Species in Bulk and at Interfaces: Interaction, Mobility, Transport, and Regulation” is derived from a series of three CECAM workshops held in 2016, 2018, and 2022. The aim of this issue is to advance our understanding of the complex phenomena surrounding charged species in various environments from a fundamental perspective, with some excursions into practical applications. The issue addresses the characterization of the interactions between a broad variety of charged species as well as of the complex macroscopic behaviour arising from these interactions. Modelling pair interactions between highly charged macromolecules and surfaces resulting from (possibly mobile) charges presents significant challenges; equally challenging is understanding how these interactions govern self-assembly, assembly kinetics, transport in dense systems, response to external electric fields, and regulatory mechanisms. In particular, the transport of charged species, both in bulk and at interfaces, has garnered significant attention due to its fundamental and technological implications. The role of electrostatics and dynamics in biological systems – spanning a wide range of length scales has become a rapidly growing area of research.
EPJ E Highlight - A metareview of active matter research
- Details
- Published on 15 April 2025
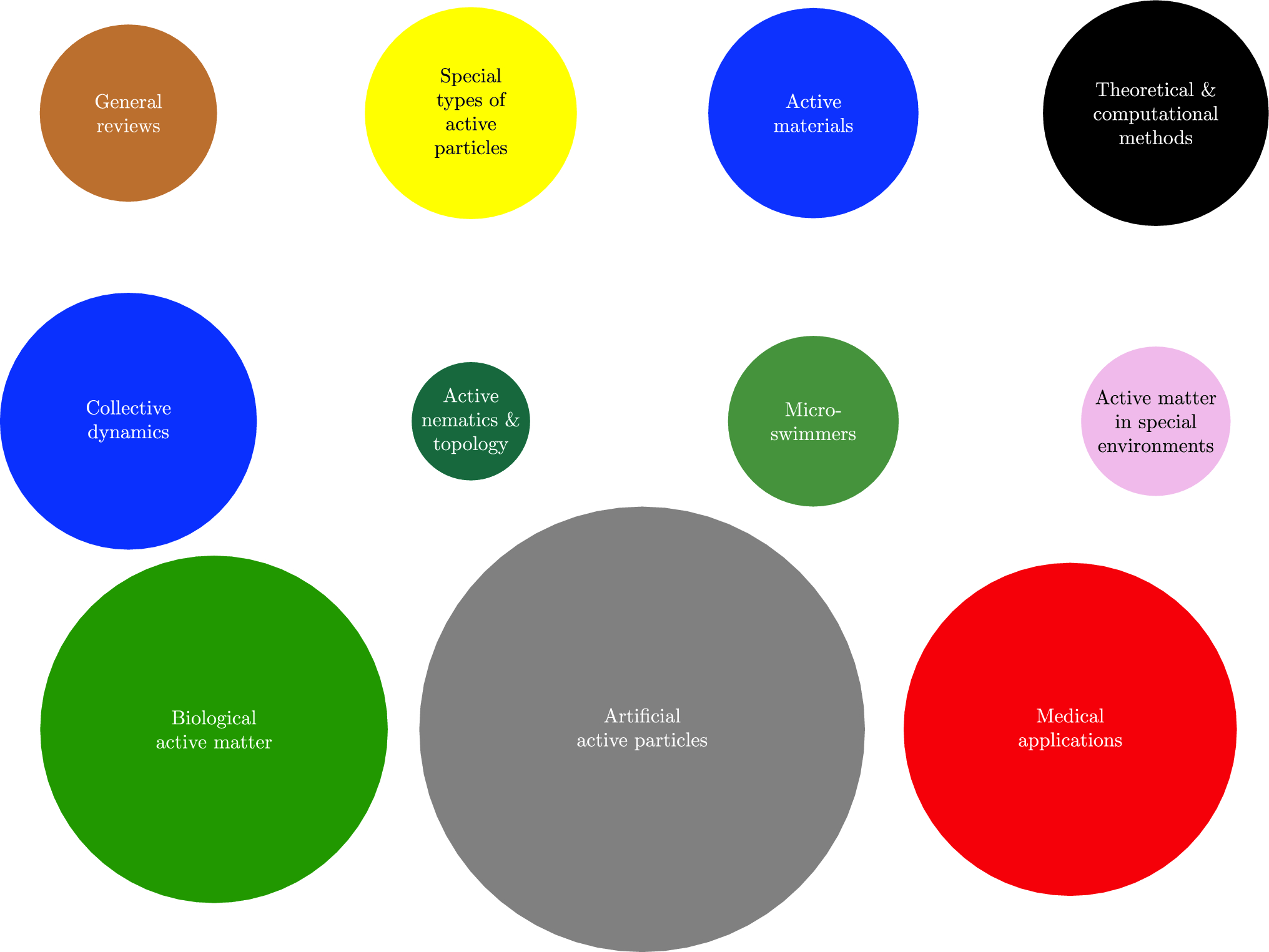
This paper presents a ‘review of reviews’ of the rapidly growing and diversifying field of active matter research, providing a valuable overview of the field as a whole
Active matter is a term describing systems of particles which harvest the energy in their surrounding environment, usually to propel themselves forward. With examples including flocks of birds, bacteria colonies, and swimming microrobots, active matter can show deeply complex behaviours, which can be exploited for real-world applications.
In recent years, researchers have shown a rapidly growing interest in active matter – with over 1,000 reviews on the topic being published to date. Yet with this growing interest, the field has been divided into an increasingly diverse range of subfields, and it is now becoming increasingly difficult to gain an overview of the field as a whole.
EPJ E Highlight - Understanding arrangements of suspended particles in reversing flows
- Details
- Published on 13 March 2025

Experiments reveal new insights into how suspended particles rearrange themselves when the flow direction of their suspending fluid is reversed
When particles are suspended in viscous fluids of the same density, their distribution becomes uneven as the fluid is subjected to straining flows. This affects the suspension’s viscosity, especially during flow reversals. Yet although this behaviour is well understood in steady, uniform flows, less is known about how the microstructures of fluid suspensions will evolve in more complex scenarios.
Through new experiments detailed in EPJ E, a team led by Irene Ippolito at the University of Buenos Aires and Georges Gauthier at the University of Paris-Saclay have uncovered new insights into this evolving structure when a suspension’s flow direction is periodically reversed within a narrow gap.
EPJ E Highlight - Exploring the impacts of particle parameters on self-propelled motions
- Details
- Published on 11 December 2024
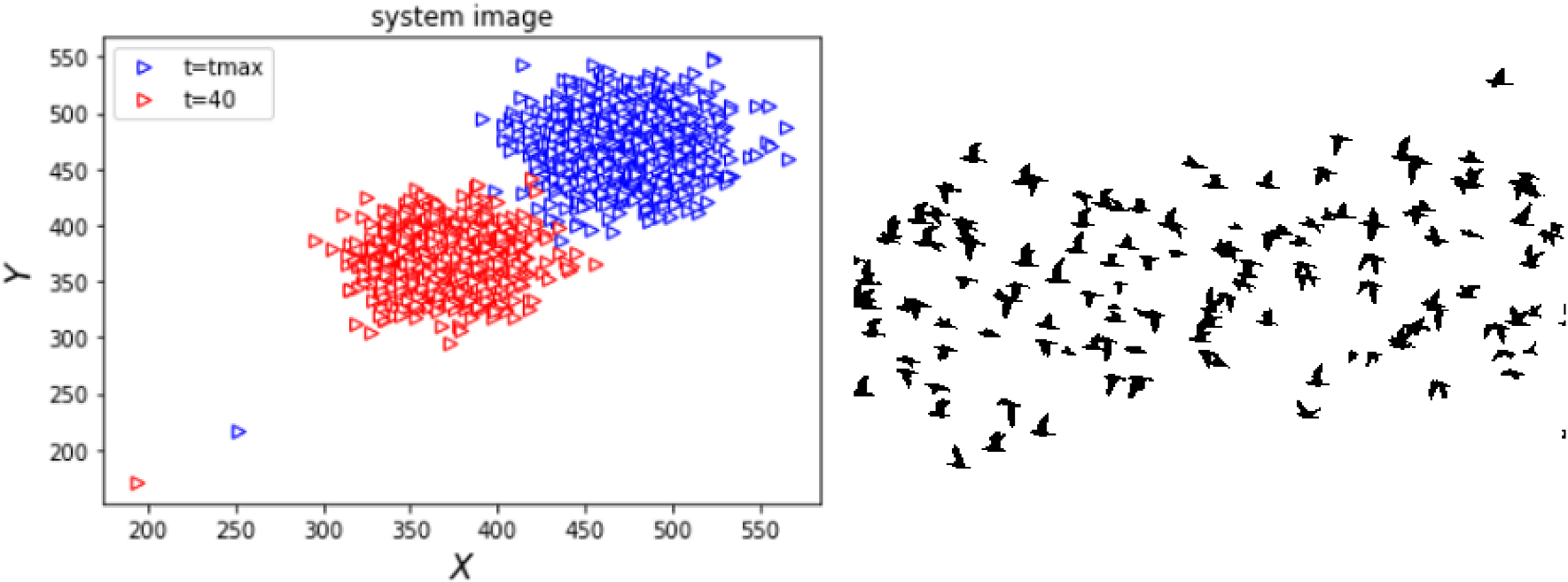
Phase transitions in the collective motions of self-propelled particles are directly impacted both by the initial velocity of each particle, and the repulsive radius surrounding them.
Collective motions of self-propelled particles can be found across many systems in nature. One of the most striking features of this phenomenon is the way in which systems transition between different states of motion: a behaviour which can be compared directly with phase transitions in physics. So far, however, it is still not fully understood how these transitions are impacted by the initial parameters of these deeply complex systems.
Through new analysis published in EPJ E, Salma Moushi and colleagues at the University of Hassan II, Morocco, show how the conditions required for transitions to occur are heavily dependent on the initial velocities of each particle, and the repulsion radius surrounding them.
EPJ E Highlight - Macroscopic and microscopic structures of clay-surfactant mixtures
- Details
- Published on 28 October 2024

As a tribute to the late colloid scientist Isabelle Grillo, two of her principal co-workers have published a summary of some of the ground-breaking work in her PhD thesis.
The premature death of Isabelle Grillo (1972-2019), a distinguished colloid scientist at the Institut Laue-Langevin (ILL), Grenoble, France, left much of the work on clay-surfactant mixtures that she had presented in her PhD thesis still unpublished. This gap has now been filled by two of her former co-workers, Sylvain Prévost of ILL and Thomas Zemb of the Institut de Chimie Separative de Marcoule, Bagnols-sur-Cèze, who have published an extensive overview of this work and her legacy in EPJ E.
EPJ E Highlight - Investigating the Flow of Fluids with Non-Monotonic, ‘S-shaped’ Rheology
- Details
- Published on 28 October 2024

Analysis of cornstarch suspensions under different stresses suggests that some fluids with non-monotonic rheology can segregate into regions of high and low viscosity in a capillary tube.
Water and oil, and some other simple fluids, respond in the same way to all levels of shear stress and, as termed Newtonian fluids, their viscosity is constant for all stresses although it will vary with temperature. Other non-Newtonian fluids exhibit much more complex patterns of behaviour under different stresses and pressure gradients. Laurent Talon and Dominique Salin from Université Paris-Sacly, Paris, France have now shown that, under certain circumstances, cornstarch suspensions can display a banding pattern with alternating regions of high and low viscosity. This work has been published in the journal EPJ E.
EPJ E Highlight - α-SAS: Improving measurements of complex molecular structures
- Details
- Published on 12 July 2024
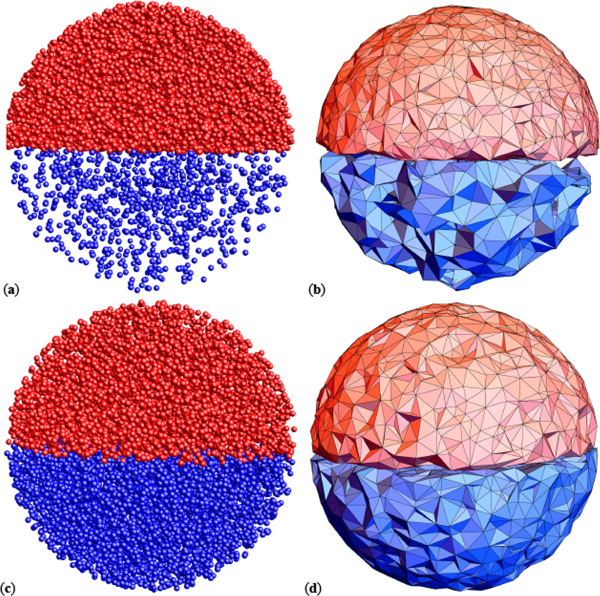
Integrating small-angle neutron scattering with machine learning algorithms could enable more accurate measurements of complex molecular structures.
Small-angle scattering (SAS) is a powerful technique for studying nanoscale samples. So far, however, its use in research has been held back by its inability to operate without some prior knowledge of a sample’s chemical composition. Through new research published in EPJ E, Eugen Anitas at the Bogoliubov Laboratory of Theoretical Physics in Dubna, Russia, presents a more advanced approach, which integrates SAS with machine learning algorithms.
EPJ E Topical Issue: Festschrift in honor of Philip (Fyl) Pincus
- Details
- Published on 26 June 2024

Guest Editors: David Andelman,
Jean-Marc Di Meglio, and Cyrus R. Safinya
This topical issue comprises 49 contributions covering a broad range of topics, which advance the understanding of soft and biological matter systems from physical and chemical aspects. More than 200 scientists globally contributed to this noteworthy Festschrift, which is divided into thematic categories.
The first theme is focused on equilibrium and non-equilibrium soft matter systems, including topics associated with polymers and colloidal systems, in uncharged and charged systems, where Pincus has made lasting contributions. In addition, other contributions are concerned with liquids, flowing and active matter, and granular systems. The second theme groups together many contributions that are focused on biological physics, including properties of the cell cytoskeleton and associated proteins, intrinsically disordered proteins, lipid membranes, membrane-associated proteins, and assembly and interactions of viral capsids with lipids and polymers. A third group of contributions is in the nascent field of biomolecular and biomimetic materials at the crossroads between physics, chemistry, bioengineering, and materials science. Finally, systems dealing with far-from-equilibrium states of matter in biology are addressed by a few contributions focusing on the physical properties of living cells.
All articles of this collection are available here and are freely accessible until 20 August 2024. For further information read the Editorial.
EPJ E Highlight - Tuning the movement of a self-propelled robot to the physics of living matter
- Details
- Published on 24 June 2024
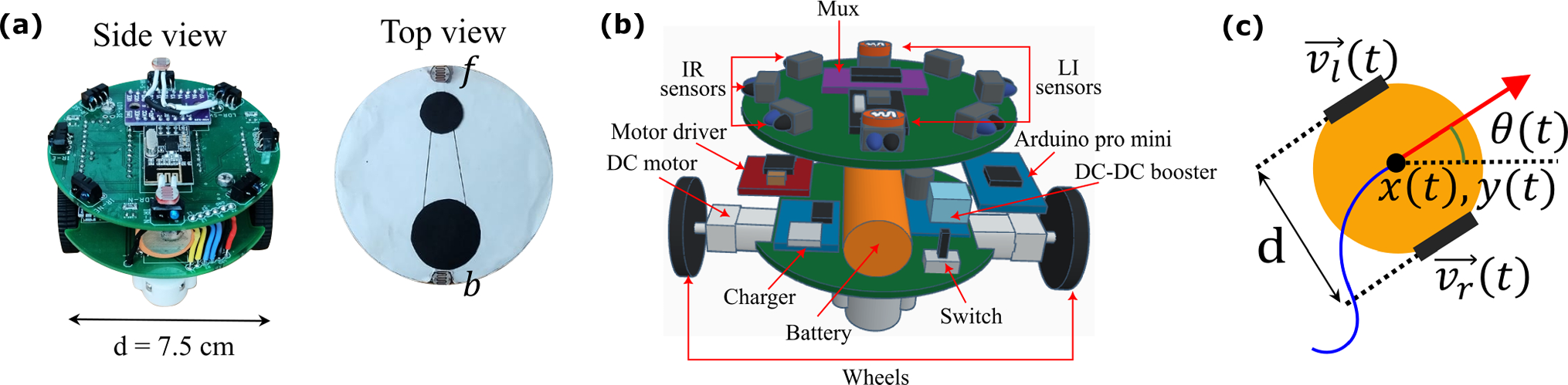
The two-wheeled robot employs a range of complex active dynamics that can be implemented with precise control.
Robots are becoming an increasingly important part of our lives, performing jobs that are too dangerous for humans. This can often involve navigating complex environments, something rigid-bodied autonomous robots find difficult. Such robots faced similar challenges when miniaturised and used to model physics of living matter.
These challenges could be countered by a robot that can move with the mobility of living things and can respond to environmental signals just like a cellular organism. To model such systems experimentally, it is necessary to develop a tunable system that can replicate life-like dynamics.
In a new paper in the EPJ E, the authors, including Nitin Kumar from the Indian Institute of Technology Bombay and his co-authors, describe the development of a scheme for generating tunable active dynamics in a self-propelled robotic device. The result is a two-wheeled robot that utilizes a simple differential-drive mechanism, enabling a range of complex active dynamics to be implemented with precise control.




